New high-throughput array greatly accelerates mouse DNA methylation characterization
July 13, 2022
GRAND RAPIDS, Mich. (July 13, 2022) — A new high-throughput genomic array offers an unprecedented look into the mouse epigenome, giving researchers an in-depth view into the basis of health and disease in a vital model organism.
The array was developed by scientists at Van Andel Institute, Children’s Hospital of Philadelphia and University of Pennsylvania in collaboration with Illumina and FOXO Technologies. It contains more than 296,000 probes that represent the diversity of murine DNA methylation biology.

“DNA methylation is one of the pillars of epigenetics research,” said VAI Associate Professor Hui Shen, Ph.D., co-corresponding author of the study that describes the array, which published today in Cell Genomics. “This array gives scientists around the world a powerful new tool to understand the role of methylation in normal processes and in diseases like cancer and many others.”
Since the 1980s, it has become clear that epigenetics plays a role in virtually every aspect of health and disease. This revelation has given rise to a growing field that has illuminated vital insights into myriad diseases such as cancer, which is now known to be driven by both genetic and epigenetic errors.

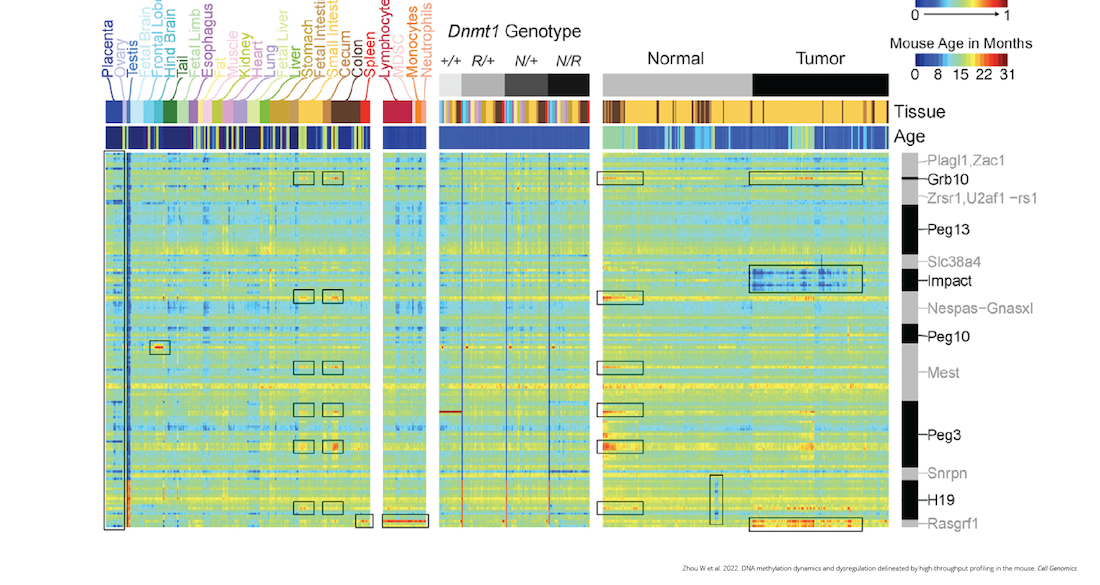
The new array includes markers for an extensive list of biological features, including genomic imprinting, aging, cancer, variably methylated regions, germ cell development, tissue specificity, X-linked probes, genomic imprinting and epigenetic clocks.
Development of the array was led by Shen, VAI Professor Peter W. Laird, Ph.D., co-corresponding author of the study, and Children’s Hospital of Philadelphia and University of Pennsylvania Assistant Professor Wanding Zhou, Ph.D., a former postdoctoral fellow in the Laird and Shen labs.

“We learned many lessons from the current and previous generations of human arrays and have improved the overall quality of probe design,” said Zhou, who also is a corresponding author of the study. “For example, we optimized the probe selection to cover the diverse biology of DNA methylation in mice while minimizing the chance of probe misuse and artifacts. The array also features probe sets that will enable comparative epigenome analysis between humans and mice as well as among other rodents.”
The array allows scientists to interrogate methylation more quickly and more deeply than previous methods. It enabled the team to develop a rich atlas of DNA methylation profiles across more than 1,200 samples, representing diverse cell types, strains and pathologies. Before now, DNA methylation data were available but not centralized. The atlas, made possible by the array, offers scientists a central repository for this critical information. Additionally, the array has been implemented in the popular bioinformatics tool SeSAMe.
As part of their validation process, the team also re-ran experiments from the early days of epigenetics research, revealing fidelity to the original findings and achieving results much faster.
“This new array is an absolutely essential tool that will provide a bridge to new breakthroughs,” said VAI Chief Scientific Officer Peter A. Jones, Ph.D., D.Sc. (hon), an internationally renowned epigenetics pioneer and a study co-author. “It allowed us to further validate work we did more than 40 years ago. Its speed, depth and efficiency will allow us to accelerate discovery by identifying genetic and epigenetic changes more quickly and in greater detail than ever before.”
Authors include Toshinori Hinoue, Ph.D., Owen Mitchell, Kelly K. Foy, Kwang-Ho Lee, Ph.D., Alexandra VanderArk, Julie M. Koeman, Manpreet Kalkat, Ph.D., Nathan J. Spix, Bryn Eagleson, J. Andrew Pospisilik, Ph.D., Piroska E. Szabó, Ph.D., Nicole A. Vander Schaaf, Ph.D., Liang Kang, Ashley K. Wiseman, Connie M. Krawczyk, Ph.D., and Marie Adams of Van Andel Institute; Waleed Iqbal, Sol Moe Lee, Ph.D., Ethan J. Moyer and Wubin Ding, Ph.D., of Children’s Hospital of Philadelphia; Bret Barnes, Ph.D., and Rishi Porecha, Ph.D., of Illumina; Marisa S. Bartolomei, Ph.D., of University of Pennsylvania; and Brian H. Chen, Ph.D., MPH, of FOXO Technologies. VAI’s Vivarium and Transgenic Core, Flow Cytometry Core, Genomics Core, and Scientific Computing and Data Management team contributed to this work.
Research reported in this publication was supported by Van Andel Institute; Children’s Hospital of Philadelphia; and the National Cancer Institute of the National Institutes of Health under award nos. R01CA157918, R01CA212374 and R01CA234125 (Laird). Shen is supported by the National Cancer Institute of the National Institutes of Health under award no. R37CA230748. Jones is supported by the National Cancer Institute of the National Institutes of Health under award no. R35CA209859. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or other funders.
